某学生取9g淀粉溶于水,为测定淀粉的水解百分率,其程序如图:
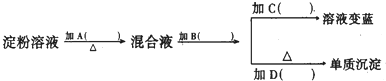
(1)各步所加试剂为:A______、B______、C______、D______.
(2)只加A溶液而不加B溶液是否可以______,理由______.
(3)当析出2.16g沉淀时,淀粉水解率为______.
(1)测定淀粉的水解完全与否,加NaOH中和H2SO4后,加银氨溶液或新制Cu(OH)2悬浊液,检验-CHO,所以淀粉水解,A为H2SO4作催化剂,加入的B为NaOH,中和掉H2SO4,因为氧化反应不能在酸性条件下发生,C为碘水,D为银氨溶液,银氨溶液和醛发生氧化还原反应生成银单质,
故答案为:H2SO4;NaOH;碘水;[Ag(NH3)2]OH;
(2)银镜反应必须在碱性条件下才能发生反应,在酸性条件下不发生反应,所以加入的氢氧化钠溶液将硫酸中和掉而使环境呈碱性;
故答案为:不可以;银镜反应必须在碱性条件下发生,所以必须加入NaOH溶液将催化剂H2SO4中和掉;
(3)(C6H10O5)n淀粉+nH2O
nC6H12O6(葡萄糖),催化剂 △
(C6H10O5)n~nC6H12O6~2nAg
162n 216ng
x2.16g
淀粉质量为:x═1.62g,
则淀粉的水解率为
×100%=18%;1.62g 9g
故答案为:18%.

