问题
选择题
已知标准状况下:①6.72L CH4 ②3.01×1023HCl ③23.1g CCl4 ④2.3g Na,下列对四种物质的
关系由小到大表示正确的是
A.体积④<①<②<③
B.密度①<④<③<②
C.原子数④<③<②<①
D.质量④<①<③<②
答案
答案:C
题目分析:①V(CH4)=6.72L,n(CH4)= 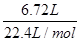 =0.3mol,m(CH4)=0.3mol×16g/mol=4.8g,ρ=
=0.3mol,m(CH4)=0.3mol×16g/mol=4.8g,ρ=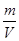 =
=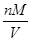 =0.714×10-3kg/L,N(原子数)=5×n(CH4)×NA/mol=1.5NA;②n(HCl)=
=0.714×10-3kg/L,N(原子数)=5×n(CH4)×NA/mol=1.5NA;②n(HCl)=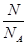 =
=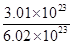 =0.5mol,V(HCl)= n(HCl)×22.4L/mol=11.2L,m(HCl)=0.5mol×36.5g/mol=18.25g,ρ=
=0.5mol,V(HCl)= n(HCl)×22.4L/mol=11.2L,m(HCl)=0.5mol×36.5g/mol=18.25g,ρ=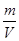 =1.62×10-3kg/L, N(原子数)=2×n(HCl)×NA/mol=0.6NA;③m(CCl4)=23.1g,n(CCl4)=
=1.62×10-3kg/L, N(原子数)=2×n(HCl)×NA/mol=0.6NA;③m(CCl4)=23.1g,n(CCl4)= 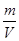 =0.15mol,N(原子数)=5×n(CCl4 )×NA/mol=0.6NA ,CCl4 标准状况下为液体,故其密度体积无法比较;④m(Na)=2.3g,n(Na)=0.1mol,N(原子数)=1×n(Na)×NA/mol="0.1" NA,因为Na为金属,故其密度体积无法比较。故C正确。
=0.15mol,N(原子数)=5×n(CCl4 )×NA/mol=0.6NA ,CCl4 标准状况下为液体,故其密度体积无法比较;④m(Na)=2.3g,n(Na)=0.1mol,N(原子数)=1×n(Na)×NA/mol="0.1" NA,因为Na为金属,故其密度体积无法比较。故C正确。
