问题
选择题
A.C+H2OCO+H2;△H=+10.94kJ/mol
B.C(s)+H2O(g)CO(g)+H2(g); △H=+10.94kJ/mol
C.C(s)+H2O(g)CO(g)+H2(g); △H=-131.3kJ/mol
D.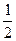 C(s)+
C(s)+ 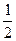 H2O(g)
H2O(g)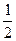 CO(g)+
CO(g)+ 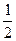 H2(g); △H=+65.64kJ/mol
H2(g); △H=+65.64kJ/mol
答案
D
A项未标明物质的聚集状态,∆H的值也不对,B、C项的∆H值均不对。
