问题
选择题
已知:
(1)Fe2O3(s) + C(s)=
C(s)= CO2(g)+2Fe(s) ΔH=+234.1 kJ/mol
CO2(g)+2Fe(s) ΔH=+234.1 kJ/mol
(2)C(s)+O2(g)=CO2(g) ΔH=-393.5 kJ/mol
则2Fe(s)+ O2(g)=Fe2O3(s) 的ΔH是( )
O2(g)=Fe2O3(s) 的ΔH是( )
A.-824.4 kJ/mol
B.-627.6 kJ/mol
C.-744.7 kJ/mol
D.-169.4 kJ/mol
答案
答案:A
题目分析:2Fe(s)+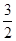 O2(g)=Fe2O3(s)可由(2)×2/3-(1)得到,所以其ΔH=-393.5×2/3-234,答案为A.
O2(g)=Fe2O3(s)可由(2)×2/3-(1)得到,所以其ΔH=-393.5×2/3-234,答案为A.
