问题
计算题
在足量浓硝酸中加入3.84gCu片,反应完全后,使产生的气体(只含有一种气体)通入800 mL H2O中充分反应,设反应后溶液体积不变。
(1)写出产生的气体通入水中的化学反应方程式,并指出氧化剂与还原剂质量之比;
(2)试求将气体通入水中充分反应后得到的溶液的物质的量浓度。
答案
解:(1)3NO2 + H2O===2HNO3 + NO; 1:2
(2)Cu + 4HNO3=== Cu(NO3)2 + 2NO2↑+ 2H2O
1 2
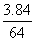 mol x = 0.12 mol
mol x = 0.12 mol
3NO2 + H2O = 2HNO3 + NO
3 2
0.12 mol y = 0.08 mol

