Q、W、X、Y、Z为五种原子序数递增的短周期元素。
已知:①Q原子核外电子数等于电子层数,且与W组成的化合物是沼气的主要成分;
②W与Y、X与Y组成的化合物是机动车排出的大气污染物;
③Y与Z能形成电子总数为30和38的两种离子化合物。
(1)W在周期表的位置:________;电子数为38的Y与Z形成化合物的电子式:________。
(2)工业合成XQ3,ΔH<0;下列措施既能加快反应速率,又能使原料转化率一定都提高的是________。
A.升高温度
B.加入高效催化剂
C.将XQ3及时分离出去
D.增大反应体系压强E.增大反应物浓度
(3)标准状况下22.4 L X的气态氢化物,与0.5 L 1 mol/L的X的最高价氧化物的水化物反应,所得碱性溶液中离子浓度由大到小顺序是_______(用化学式表示)。
(1)第二周期ⅣA族 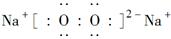
(2)D (3)c(NH4+)>c(NO3-)>c(OH-)>c(H+)
由①推知Q为H,沼气的主要成分是甲烷,故W为C;由②中W与Y形成的化合物是机动车排出的大气污染物可知,该化合物为CO,故Y为O,Q、W、X、Y原子序数递增,故X是N;Na2O含有30个电子,Na2O2含有38个电子,故Z为Na。(1)W是碳元素,为元素周期表中第6号元素,位于第二周期ⅣA族;Na2O2为离子化合物,且存在过氧键(—O—O—),因此其电子式为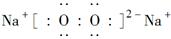 。(2)XO3为NH3,工业合成氨利用氢气与氮气反应,该反应为放热反应,升高温度可以提高反应速率,但会降低反应物的转化率,A项错;加入催化剂,可以提高反应速率,但不能提高反应物的转化率,B项错;将氨气及时分离出去可以提高原料的转化率,但同时也降低了反应速率,C项错;增大反应体系压强,体系中的物质浓度增大,反应速率增大,且该反应为气体分子数减小的反应,故也能提高反应物的转化率,D项正确;若增大一种反应物的浓度可以提高反应速率,但会降低该反应物的转化率,E项错。(3)氨气与硝酸反应的化学方程式为NH3+HNO3=NH4NO3。氨气的物质的量n(NH3)=22.4 L÷22.4 L/mol=1 mol,n(HNO3)=0.5 L×1 mol/L=0.5 mol。由此可知氨气过量0.5 mol,溶液中存在NH3·H2O的电离和NH4+的水解,因最终溶液呈碱性,故离子浓度大小顺序为c(NH4+)>c(NO)>c(OH-)>c(H+)。
。(2)XO3为NH3,工业合成氨利用氢气与氮气反应,该反应为放热反应,升高温度可以提高反应速率,但会降低反应物的转化率,A项错;加入催化剂,可以提高反应速率,但不能提高反应物的转化率,B项错;将氨气及时分离出去可以提高原料的转化率,但同时也降低了反应速率,C项错;增大反应体系压强,体系中的物质浓度增大,反应速率增大,且该反应为气体分子数减小的反应,故也能提高反应物的转化率,D项正确;若增大一种反应物的浓度可以提高反应速率,但会降低该反应物的转化率,E项错。(3)氨气与硝酸反应的化学方程式为NH3+HNO3=NH4NO3。氨气的物质的量n(NH3)=22.4 L÷22.4 L/mol=1 mol,n(HNO3)=0.5 L×1 mol/L=0.5 mol。由此可知氨气过量0.5 mol,溶液中存在NH3·H2O的电离和NH4+的水解,因最终溶液呈碱性,故离子浓度大小顺序为c(NH4+)>c(NO)>c(OH-)>c(H+)。
